A professor from RUDN University developed new liquid crystals
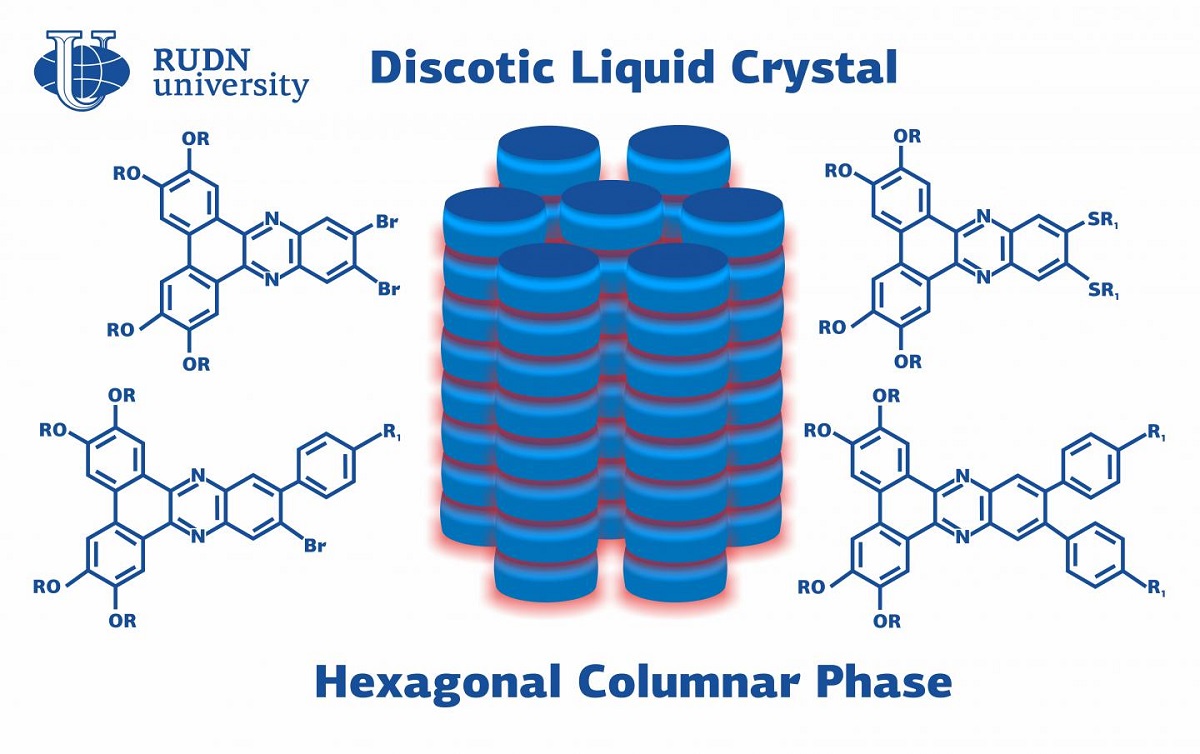
Liquid crystals are an intermediate phase between a liquid and a solid body. They are ordered like regular chrystals but at the same time have a flow like liquids. It is this duality that allows them to be used in organic LEDs and LCDs. Unlike other liquid crystals, discotic ones (DLC) are capable of self-assembly into ordered structures. This makes them a promising material for industrial electronics, namely, for the production of displays. A professor from RUDN University together with his Indian colleagues synthesized and described new dibenzophenazine-based DLCs.
“Discotic liquid crystals are interesting because of their ability to form self-assembled ordered columnar structures. In such structures, an electric charge can move along the column, which makes them useful for optoelectronic devices such as organic LEDs, organic field-effect transistors (OFET), photoelectric solar elements, and sensors,” said Prof. Viktor Belyaev, a Ph.D. in Technical Sciences from the Department of Mechanics and Mechatronics at RUDN University.
DLCs consist of disc-shaped molecules aligned in columns. In the center of each disc, there is an aromatic ring (a cyclical organic fragment) surrounded by chains of other organic fragments. Due to this aromatic center, a DLC can transfer a charge along the axis of a column. Prof. Belyaev developed discotic liquid crystals with an aromatic compound called dibenzophenazine in the center. As for the chains that surrounded it, the team tried three different types of fragments. The molecular structure of the new DLCs was studied using spectral, X-ray diffraction, and elementary analysis. Then, the team tested the three groups of DLCs in a set of experiments.
The experiments showed that alkoxy thiol chains increased the polarity of the molecules in liquid crystals thus improving the internal structure of the columns and making them more even. All new DLCs were able to withstand temperatures up to 330?. However, the crystals that consisted of smaller molecules (i.e. the ones with their aromatic center surrounded by alkyl thiols) lost their intermediary status and transitioned from the liquid crystal to the liquid form at lower temperatures (55.1 ?) that the crystals from the other two groups. This is due to the size of the molecules in the columns: the bigger they are, the more stable is the liquid crystal state.
“The new discotic liquid crystals could play an important role in organic optoelectronic devices and solar panels,” added Prof. Viktor Belyaev from RUDN University.
The results of the study were published in the Journal of Molecular Liquids.
In the last five years, academic staff of RUDN alongside the Chinese universities and research centers has achieved impressive results. More than 1,000 scientific publications were made jointly in a wide spectrum from engineering and technology to medicine and social sciences. 19 of these materials were published in top-rated journals which verifies that the works were world-class level.
RUDN University has been included among the leaders in the BRICS ESG University Ranking among 65 leading universities. They were assessed on 20 criteria, ranging from education and research to environmental and social agendas.
At a meeting of the new RUDN University Dissertation Council in the specialties of 2.1.12 “Architecture of Buildings and Structures. Creative Concepts of Architectural Activity” and 2.1.11 “Theory and History of Architecture, Restoration and Reconstruction of Historical and Architectural Heritage”, three postgraduate students — Natalia Kalinina, Evgeny Ogienko, and Yulia Loginova — were awarded PhD degrees in Architecture for the first time in the university's history.