RUDN University chemist synthesized a coordination polymer iron with a derivative of nicotinic acid
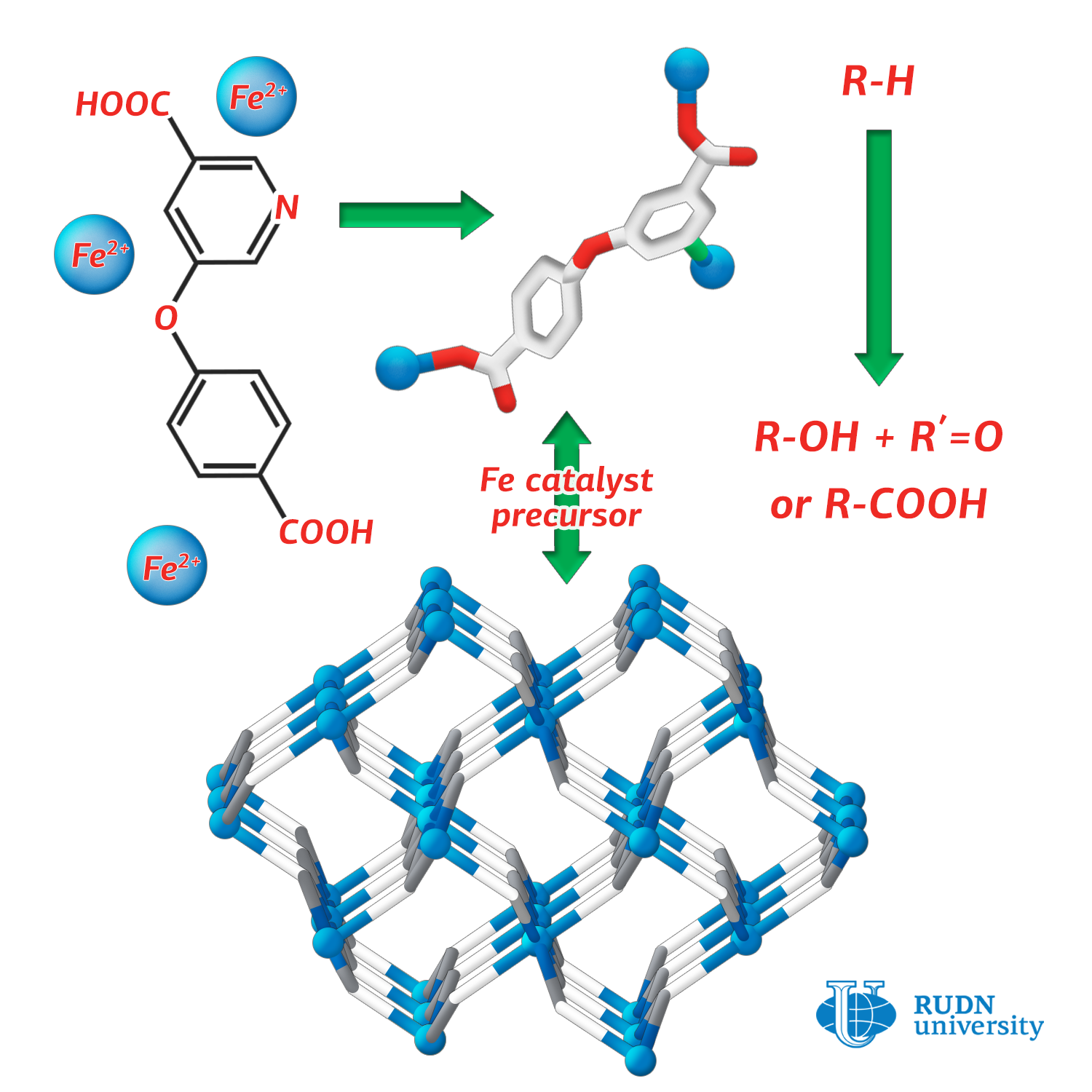
Coordination polymers are compounds that consist of a metal atom and surrounding organic ligands. They are often more stable than pure organic substances. RUDN chemist Alexander Kirillov used a nicotinic acid derivative (H2cpna) as a building block in the synthesis, and iron atoms played the role of the metal center. Substituted nicotinic acid can act as a ligand — it contains one phenyl and one pyridine ring, which are linked by an ether functional group.
To synthesize the polymer, the reaction was performed under hydrothermal conditions between iron (II) sulfate in water and H2cpna at 160 ⁰C. The synthesis lasted three days. X-ray diffraction analysis and other methods were used to confirm the structure and characteristics of the obtained substance.
The catalytic activity of the substance for different reactions was studied. Alexander Kirillov from RUDN conducted the processes of oxidation and carboxylation of propane and cyclic alkanes (saturated hydrocarbons closed in a cycle) under mild conditions. The reaction yield was 23%. For comparison, in the industrial process of oxidation of cyclohexane to cyclohexanol and cyclohexanone (products that are used in the production of plastics), the yield is only 5-10%.
The study of the catalytic activity of the polymer obtained by RUDN chemists showed that it can be used to catalyze the processes of oxidative functionalization of saturated hydrocarbons, and provide a greater yield for the reaction under mild conditions.
Article in Crystals magazine.
Imagine a world where everyone has enough food, clean water, access to education, and decent work. A world where nature is protected and the future of our planet is cared for. These are the Sustainable Development Goals—to achieve a sustainable future for all! To this end, in 2015, the United Nations (UN) defined 17 Sustainable Development Goals (SDGs). The SDGs are a global plan that helps countries and people work together towards a better future. All 193 UN member states have joined the plan.
Researchers from the Faculty of Artificial Intelligence at RUDN University conducted a large-scale study that revealed systemic errors in large language models (LLMs) when diagnosing depression based on text. This work, carried out in collaboration with colleagues from AIRI, Federal Research Center “Computer Science and Control” of the Russian Academy of Sciences, Ivannikov Institute for System Programming of the Russian Academy of Sciences, Moscow Institute of Physics and Technology, and MBZUAI, not only identifies the problem but also lays the foundation for the creation of more reliable and secure tools for detecting depression and anxiety.
Alexandra Sentyabreva, a junior researcher at the Laboratory of Cell Technologies and Tissue Engineering at RUDN Research Institute of Molecular and Cellular Medicine at the Russian University of People's Friendship, won the competition for young scientists at the All-Russian Scientific Conference “Topical Issues of Morphogenesis in Norm and Pathology.” She was awarded the Academician A.P. Avtsyn Prize.
Imagine a world where everyone has enough food, clean water, access to education, and decent work. A world where nature is protected and the future of our planet is cared for. These are the Sustainable Development Goals—to achieve a sustainable future for all! To this end, in 2015, the United Nations (UN) defined 17 Sustainable Development Goals (SDGs). The SDGs are a global plan that helps countries and people work together towards a better future. All 193 UN member states have joined the plan.
Researchers from the Faculty of Artificial Intelligence at RUDN University conducted a large-scale study that revealed systemic errors in large language models (LLMs) when diagnosing depression based on text. This work, carried out in collaboration with colleagues from AIRI, Federal Research Center “Computer Science and Control” of the Russian Academy of Sciences, Ivannikov Institute for System Programming of the Russian Academy of Sciences, Moscow Institute of Physics and Technology, and MBZUAI, not only identifies the problem but also lays the foundation for the creation of more reliable and secure tools for detecting depression and anxiety.
Alexandra Sentyabreva, a junior researcher at the Laboratory of Cell Technologies and Tissue Engineering at RUDN Research Institute of Molecular and Cellular Medicine at the Russian University of People's Friendship, won the competition for young scientists at the All-Russian Scientific Conference “Topical Issues of Morphogenesis in Norm and Pathology.” She was awarded the Academician A.P. Avtsyn Prize.