RUDN University chemist has found a way to improve solar cells
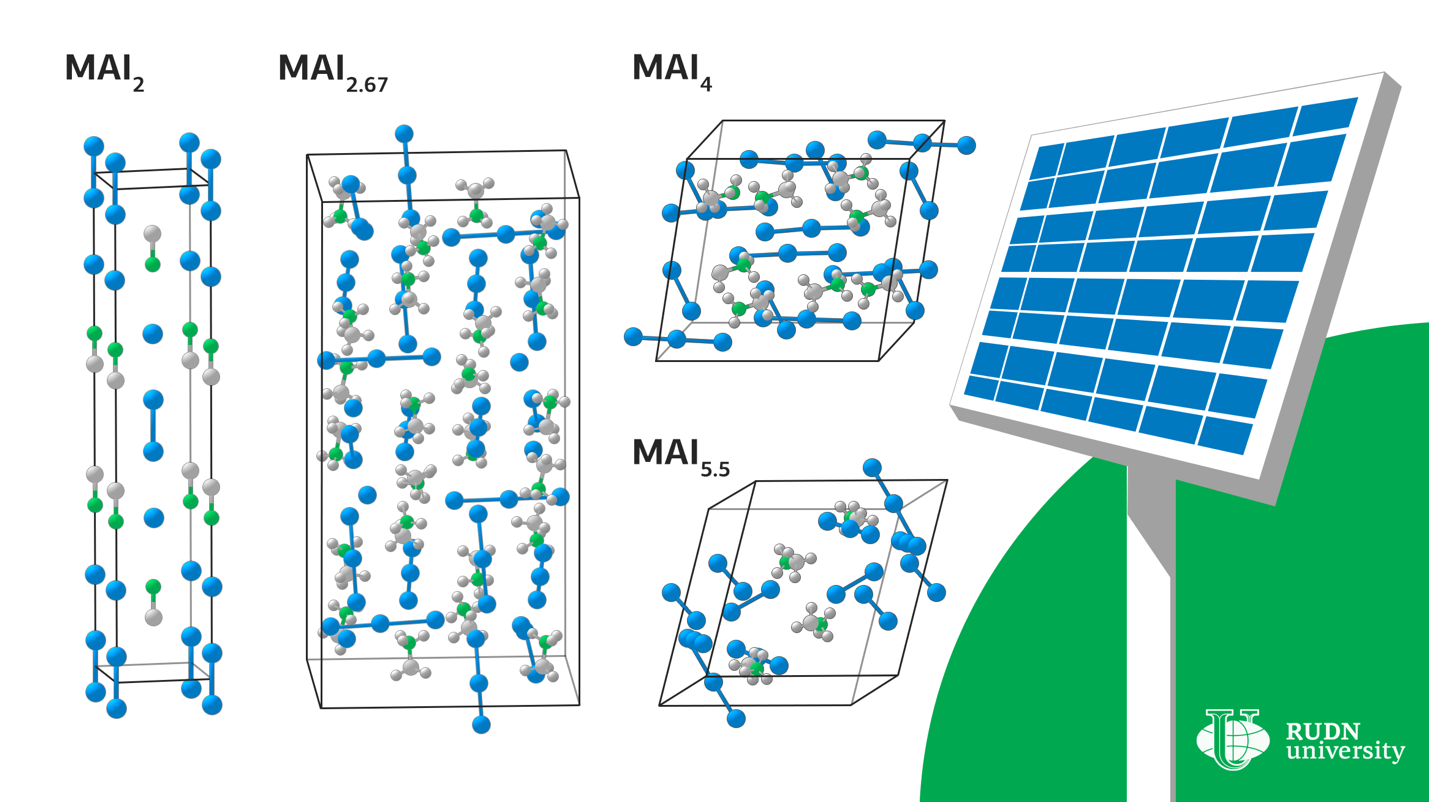
Lead-based hybrid perovskites are used in modern solar cells as a light-absorbing layer. But they are unstable to moisture, and existing technologies require the use of solutions and toxic solvents. This complicates the technology and makes it potentially dangerous. The solution to the problem can be solvent-free methods, that is, the use of melts rather than solutions — for example, you can apply a polyiodide melt to a thin film of metallic lead. However, there are few reliable studies of polyiodide chemistry. RUDN chemists investigated the properties of methylammonium (CH3NH3) and iodine compounds to find variants of compounds suitable for use in the production of perovskite solar cells.
Compounds of the methylammonium iodide (MAI) and iodine system melt at room temperature and form ionic liquids — melts that are composed exclusively of ions. The use of such liquids, which can be evenly applied to large surfaces, as precursors will bring closer the industrial production of modular solar cells based on hybrid perovskites on an industrial scale.
Already studied polyiodide-based liquids melt at room temperature only in the presence of large organic cations in the composition. RUDN chemist Viktor Khrustalev explained this difference by the fact that the methylammonium cation has a large dipole moment and is capable of forming a large number of hydrogen bonds. For small cation sizes, this leads to increased entropy during melting, which in turn lowers the melting point.
Under certain conditions, crystals of various compositions begin to fall out of the liquid-MAI2, MAI2.67, MAI4 and MAI5.5. In order to determine under what conditions the melt exists, scientists performed calculations of the enthalpy and entropy of the formation of these crystals. For all compounds except MAI2, the reaction of obtaining a compound from iodine and a composition with a lower iodine content proceeded solely due to the entropy contribution. RUDN chemists explained that the increase in enthalpy during the transition to compounds with a higher iodine content is due to the weakening of the interaction of cations with anions due to the distribution of a small negative charge on a large polyiodide anion. A similar increase in entropy is due to the complexity of polyanions and the weakening of the bonds between them.
These thermodynamic data made it possible to refine and generalize the experimental values of the boundaries in which the melt can exist.
Chemists have also found that similar effects occur in similar compounds with the formamidinium cation (FA+ = HC (NH2)2) and polybromides. Moreover, the mixed composition (MABr3) 0.15 (FAI3) 0.85 exhibits the properties of an ionic liquid from −40 to 80 °C. Such a low melting point of the precursor is favorable for the production of thin films of mixed hybrid perovskites, which exhibit maximum light-absorbing properties.
The article was published in Article in The Journal of Physical Chemistry Letters.
Imagine a world where everyone has enough food, clean water, access to education, and decent work. A world where nature is protected and the future of our planet is cared for. These are the Sustainable Development Goals—to achieve a sustainable future for all! To this end, in 2015, the United Nations (UN) defined 17 Sustainable Development Goals (SDGs). The SDGs are a global plan that helps countries and people work together towards a better future. All 193 UN member states have joined the plan.
Researchers from the Faculty of Artificial Intelligence at RUDN University conducted a large-scale study that revealed systemic errors in large language models (LLMs) when diagnosing depression based on text. This work, carried out in collaboration with colleagues from AIRI, Federal Research Center “Computer Science and Control” of the Russian Academy of Sciences, Ivannikov Institute for System Programming of the Russian Academy of Sciences, Moscow Institute of Physics and Technology, and MBZUAI, not only identifies the problem but also lays the foundation for the creation of more reliable and secure tools for detecting depression and anxiety.
Alexandra Sentyabreva, a junior researcher at the Laboratory of Cell Technologies and Tissue Engineering at RUDN Research Institute of Molecular and Cellular Medicine at the Russian University of People's Friendship, won the competition for young scientists at the All-Russian Scientific Conference “Topical Issues of Morphogenesis in Norm and Pathology.” She was awarded the Academician A.P. Avtsyn Prize.
Imagine a world where everyone has enough food, clean water, access to education, and decent work. A world where nature is protected and the future of our planet is cared for. These are the Sustainable Development Goals—to achieve a sustainable future for all! To this end, in 2015, the United Nations (UN) defined 17 Sustainable Development Goals (SDGs). The SDGs are a global plan that helps countries and people work together towards a better future. All 193 UN member states have joined the plan.
Researchers from the Faculty of Artificial Intelligence at RUDN University conducted a large-scale study that revealed systemic errors in large language models (LLMs) when diagnosing depression based on text. This work, carried out in collaboration with colleagues from AIRI, Federal Research Center “Computer Science and Control” of the Russian Academy of Sciences, Ivannikov Institute for System Programming of the Russian Academy of Sciences, Moscow Institute of Physics and Technology, and MBZUAI, not only identifies the problem but also lays the foundation for the creation of more reliable and secure tools for detecting depression and anxiety.
Alexandra Sentyabreva, a junior researcher at the Laboratory of Cell Technologies and Tissue Engineering at RUDN Research Institute of Molecular and Cellular Medicine at the Russian University of People's Friendship, won the competition for young scientists at the All-Russian Scientific Conference “Topical Issues of Morphogenesis in Norm and Pathology.” She was awarded the Academician A.P. Avtsyn Prize.