RUDN University chemist created new catalysts for click reactions
Click chemistry methods are used to synthesize libraries of substances with high chemical diversity, which is important when developing new drugs. These reactions are necessary for introduction of labels (for example, fluorescent ones) into biological macromolecules, proteins, and DNA molecules. This is used in biological and medical research.
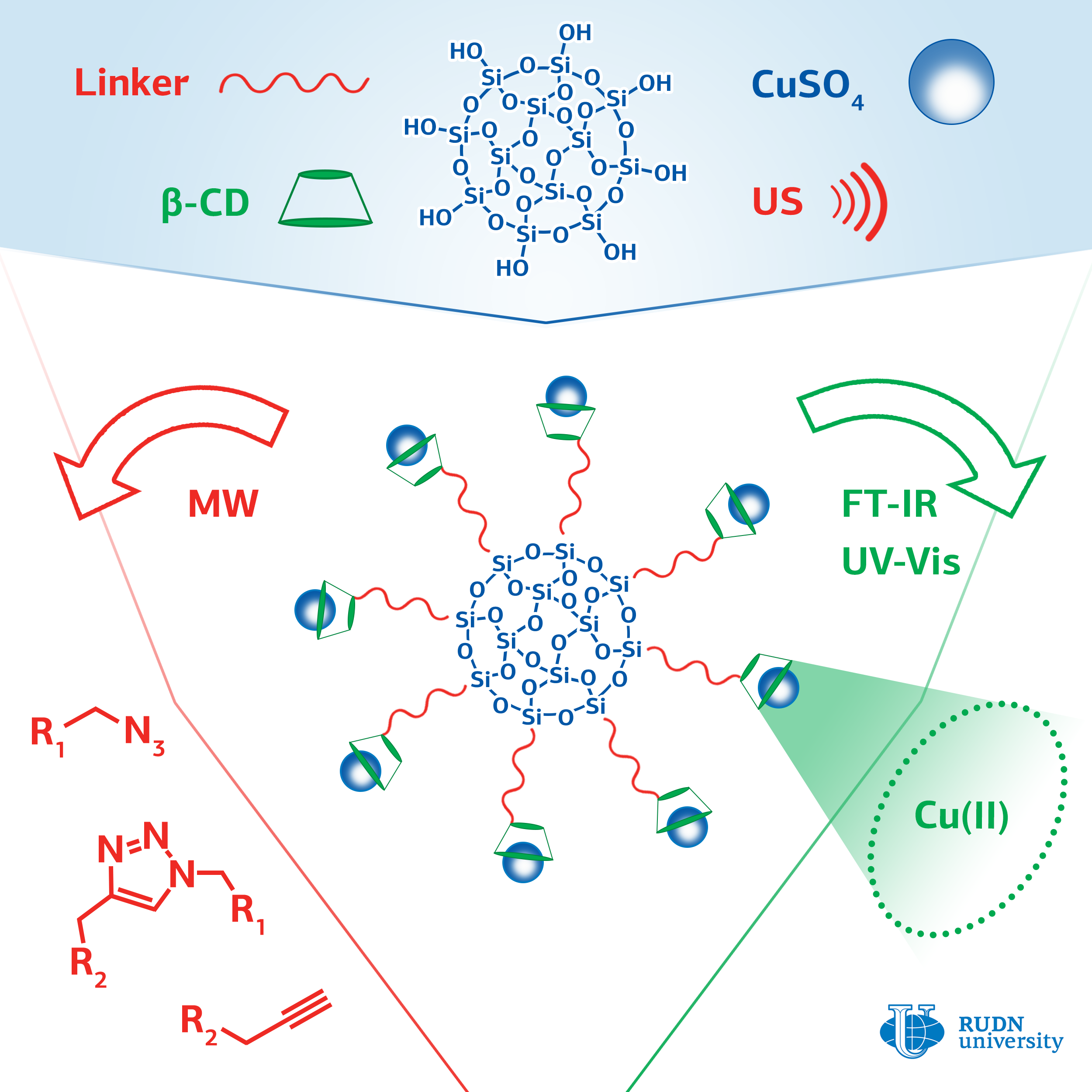
The most commonly used click chemistry reaction is the addition of a substance that contains a carbon-carbon triple bond (alkine) to a compound containing a fragment with three nitrogen atoms in a row (azide). The classic version of the reaction involves the use of copper in oxidation state (I) as a catalyst. For this, ions of copper (II) and an excess of a reducing agent are introduced into the reaction, or copper (I) is used and the reaction is conducted with protection against oxygen, which imposes certain restrictions on the application of this reaction.
A chemist from RUDN University Rafael Luque and his colleagues have developed a series of catalysts with copper ions attached to the surface of silica gel particles using cyclic cyclodextrin oligosaccharide. Cyclodextrin consists of seven glucose molecules closed in a cycle. Inside the cycle there is a container that can hold the copper ion and increase its catalytic activity. Ultrasound irradiation was used to facilitate the binding of cyclodextrin to the surface of silica gel.
The effectiveness of the created catalysts was evaluated on a model reaction of phenylacetylene with benzylazide. The researchers managed to achieve a yield of the reaction product of more than 99%. The yield with copper (II) acetate was 14%, and in the case of copper (II) sulfate, the reaction did not occur at all. The method for producing the catalyst is simple, safe for the environment, and cheap; its use does not require to add reducing agents or oxygen-free conditions. The catalysts can find application in the pharmaceutical industry and in biomedical research.
The paper was published in the journal Molecules.
In the last five years, academic staff of RUDN alongside the Chinese universities and research centers has achieved impressive results. More than 1,000 scientific publications were made jointly in a wide spectrum from engineering and technology to medicine and social sciences. 19 of these materials were published in top-rated journals which verifies that the works were world-class level.
RUDN University has been included among the leaders in the BRICS ESG University Ranking among 65 leading universities. They were assessed on 20 criteria, ranging from education and research to environmental and social agendas.
At a meeting of the new RUDN University Dissertation Council in the specialties of 2.1.12 “Architecture of Buildings and Structures. Creative Concepts of Architectural Activity” and 2.1.11 “Theory and History of Architecture, Restoration and Reconstruction of Historical and Architectural Heritage”, three postgraduate students — Natalia Kalinina, Evgeny Ogienko, and Yulia Loginova — were awarded PhD degrees in Architecture for the first time in the university's history.