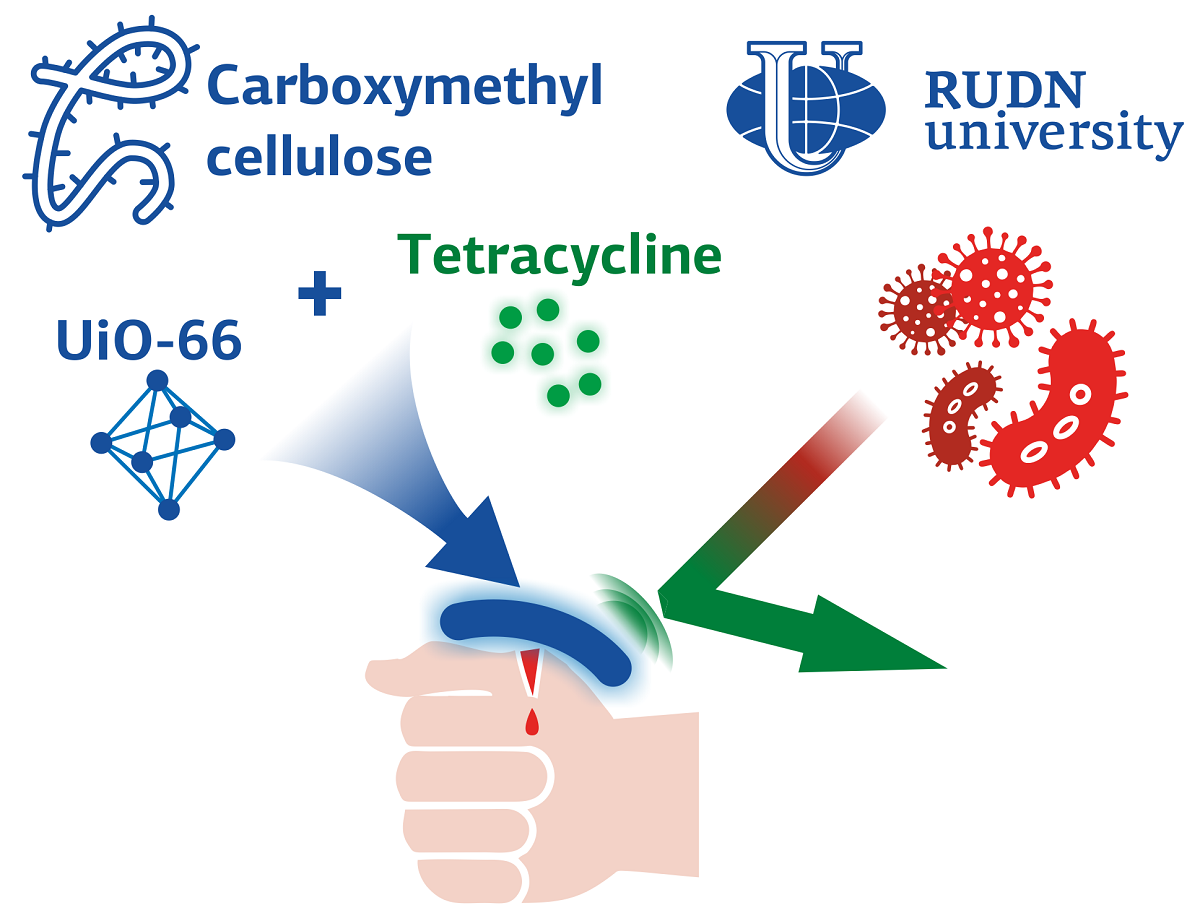
Scientists report new hydrogel to protect wounds from germs
The main aim in treating wounds is to prevent infection. With an open wound, microbes gain direct access inside the body. This significantly complicates wound healing and can cause more serious consequences, so it is important to develop antibacterial coatings for wounds. One of the modern approaches to this is hydrogel films. Unlike traditional remedies, hydrogels mimic healthy tissue and cover the wound, protecting it from germs. They also help to cool the wound and not over-dry it. In addition, hydrogels can contain a medicine that will provide the wound with additional protection. A RUDN University and SBU chemist with colleagues from Iran created such a coating and proved its effectiveness.
“Since the hydrogels’ hydrated molecular and porous structure, they can mimic the biological and physicochemical characteristics of the tissue environment hydrogel-based wound dressings have remarkable features such as providing a cooling sensation, sustaining a wet circumstance at the interfacing wound, maintaining a barrier to microorganisms, permitting gaseous exchange, and permitting wound exudate absorption,” said Ahmad Shaabani, a leading researcher at the Joint Institute for Chemical Research of RUDN and SBU.
Chemists have created a coating with pores that can contain the medicine. The film itself covers the wound, and the medicine gradually comes out of the pores, preventing microbes from spreading. The hydrogel consists of carboxymethylcellulose (a derivative of ordinary cellulose) and an organometallic framework UiO-66, with the porous structure where they put a popular antibiotic tetracycline. Scientists investigated how different concentrations of UiO-66 affect the properties of the hydrogel and its ability to release an antibiotic. Having selected the optimal parameters, chemists determined how the hydrogel with an antibiotic cope with Escherichia coli and Staphylococcus aureus.
The hydrogel showed high efficiency against both bacteria and was harmless to human connective tissue cells. In a Petri dish with a medium of artificial sweat and wound exudate, Escherichia coli and Staphylococcus aureus died within a radius of 6.5-8.5 mm from the drug. Initially, there was a high rate of antibiotic release (up to 50% of the drug came out of the hydrogel in the first five hours), then the rate stabilized, and the hydrogel slowly released the remaining drug over the next three days.
“Designing an antibacterial agent with a suitable water vapour permeability, good mechanical properties, and controlled antibiotic release is a promising method for stopping bacterial infection in wound tissue. The designed hydrogel films can attract great attention to construct a safe and straightforward system through a green method and control the microbial growth in wound tissue,” said Ahmad Shaabani, a leading researcher at the Joint Institute for Chemical Research of RUDN and SBU.
The results are published in the International Journal of Biological Macromolecules.
In the last five years, academic staff of RUDN alongside the Chinese universities and research centers has achieved impressive results. More than 1,000 scientific publications were made jointly in a wide spectrum from engineering and technology to medicine and social sciences. 19 of these materials were published in top-rated journals which verifies that the works were world-class level.
RUDN University has been included among the leaders in the BRICS ESG University Ranking among 65 leading universities. They were assessed on 20 criteria, ranging from education and research to environmental and social agendas.
At a meeting of the new RUDN University Dissertation Council in the specialties of 2.1.12 “Architecture of Buildings and Structures. Creative Concepts of Architectural Activity” and 2.1.11 “Theory and History of Architecture, Restoration and Reconstruction of Historical and Architectural Heritage”, three postgraduate students — Natalia Kalinina, Evgeny Ogienko, and Yulia Loginova — were awarded PhD degrees in Architecture for the first time in the university's history.