RUDN University chemists discovered the mechanism of radiation instability of lithium tetraborate
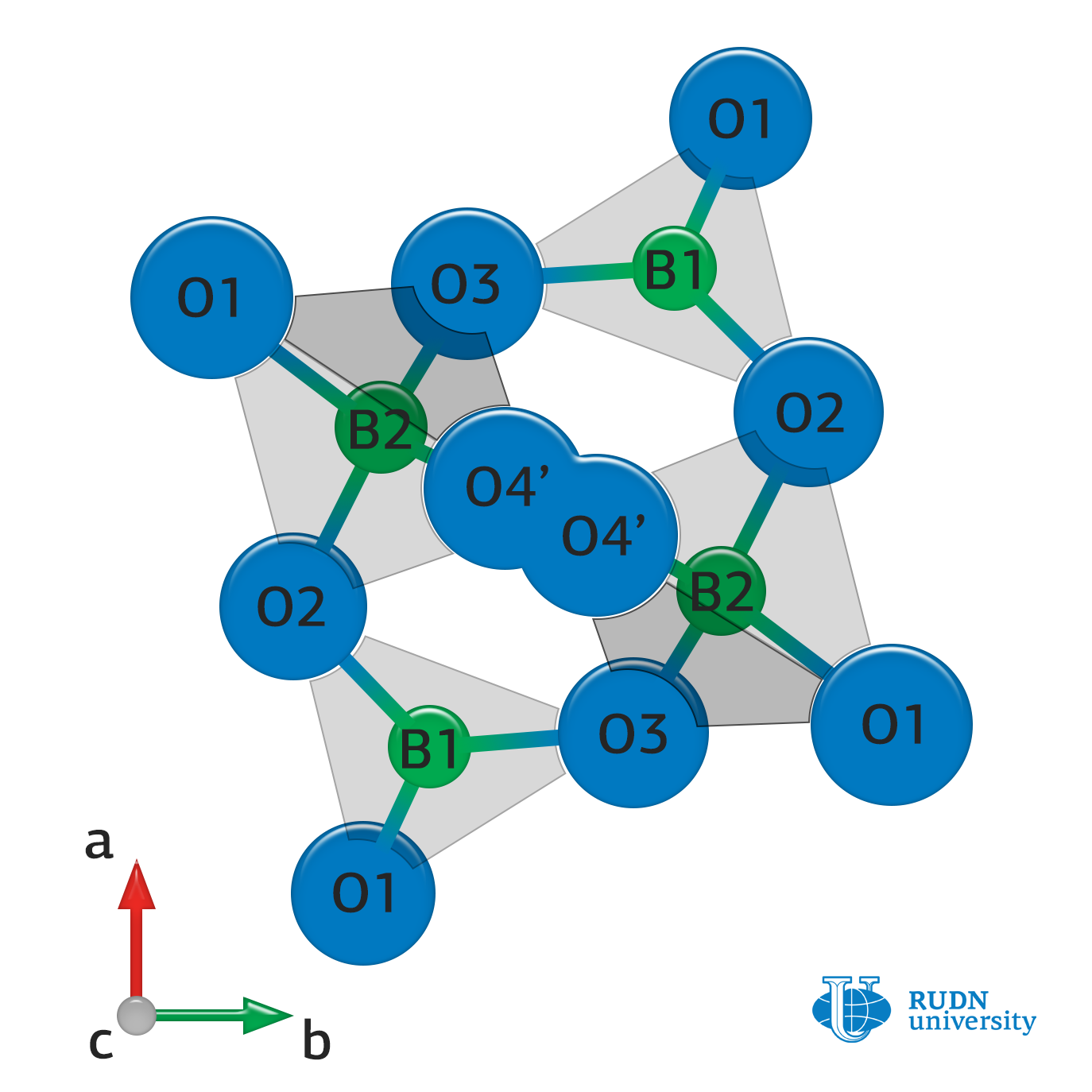
Lithium tetraborate was the first material thermoluminescent radiation dosimeters, which were highly sensitive to x-ray, gamma, and beta radiation, were made of. When ionising radiation enters a thermoluminescent dosimeter, the latter “stores” the absorbed energy due to the transfer of electrons to higher energy levels. When heated above a certain temperature, the electrons emit previously absorbed energy, and the dosimeter begins to glow. The light intensity is proportional to the amount of absorbed radiation. In order to make lithium tetraborate capable of this, impurities of manganese, silver, or other metals are introduced into it, which act as traps for those electrons that were excited by ionising radiation. But because of these impurities, the radiation resistance of the substance decreases. It has not been known why, until now.
RUDN University chemist Alexander Zubov and his colleagues compared ceramic samples based on lithium tetraborate with impurities of manganese, copper, zinc, tin, and beryllium. It turned out that the radiation stability of the substance is deteriorating due to the rupture of chemical bonds in the boron-oxygen network. And while the boron-oxygen lattice in a pure substance is capable of restoring itself during heating, the introduction of manganese interferes with this process.
The more evenly manganese is distributed in the structure of lithium tetraborate, the less negative impact it has on the radiation stability of the material is. Copper and tin prevent the clustering of manganese, forming bound complexes with it, thereby preventing it from “migrating” and “sticking” to the crystal lattice during recharging of the dosimeter. Moreover, ceramics with addition of tin, unlike of copper, also has thermoluminescent properties that allow its effective use in dosimetry.
Understanding of the physicochemical processes that occur during irradiation of a material is necessary to create new radiation resistant materials. The RUDN University chemists were able not only to explain the mechanism of radiation destruction of lithium tetraborate, but also to apply the new knowledge to create a material with a better composition, which can later be used in advanced pocket radiation dosimeters. In addition, the authors argue that their experimental approach, which involves searching for clustered manganese in the structure of lithium tetraborate, can be used as a new effective way to certify the radiation resistance of thermoluminescent dosimeters.
The work was published in the journal Radiation Measurements.
Products derived from microalgae represent a cutting-edge development in the field of bioeconomy. The potential of this biological resource was discussed at the international research seminar “Foundations for a Green Sustainable Energy”, part of the BRICS Network University’s thematic group on “Energy”. The event was organized by the Institute of Ecology at RUDN University.
Ambassadors of Russian education and science met at a conference in RUDN University to discuss how they can increase the visibility of Russian universities and research organizations in the world, and attract more international students in Russia.
The international scientific seminar hosted by RUDN Institute of Ecology “Experience of participation in student organizations as a way to form career skills” united scholarship recipients of the International Student Mobility Awards 2024 and Open Doors, along with members of the scientific student society “GreenLab” and the professional student association “Kostyor (Bonfire)” shared their projects focused on environmental protection.