RUDN University chemist creates a catalyst to remove paracetamol from wastewater
The widespread use of medicines and hygiene products has become one of the main factors of wastewater pollution. Imperceptible “treatment” with drugs that can enter the human body from insufficiently purified water poses a serious threat to human health and the environment. Biodegradation is usually employed for wastewater treatment when the job is done by microorganisms. The second option is adsorption, that is, the adsorption of harmful substances by filters-adsorbents. But in this case, it is necessary to solve the issue of recycling filters. Ozone oxidation and some other oxidation methods are also used, but they are expensive and require special equipment.
However, many harmful substances are not biodegradable. One such substances is acetaminophen, better known to consumers as paracetamol or panadol. It is the world's most common antipyretic and painkiller, which is sold over the counter in many countries. About 58-68 percent of the paracetamol in the body is excreted through the kidneys in urine and enters the city sewers. In European countries, scientists estimate that the concentration of acetaminophen in wastewater that has already been treated, can reach 6 micrograms per liter, and in the United States – 10 micrograms per liter. Therefore, the search for effective and affordable ways to remove this substance from wastewater is an urgent task.
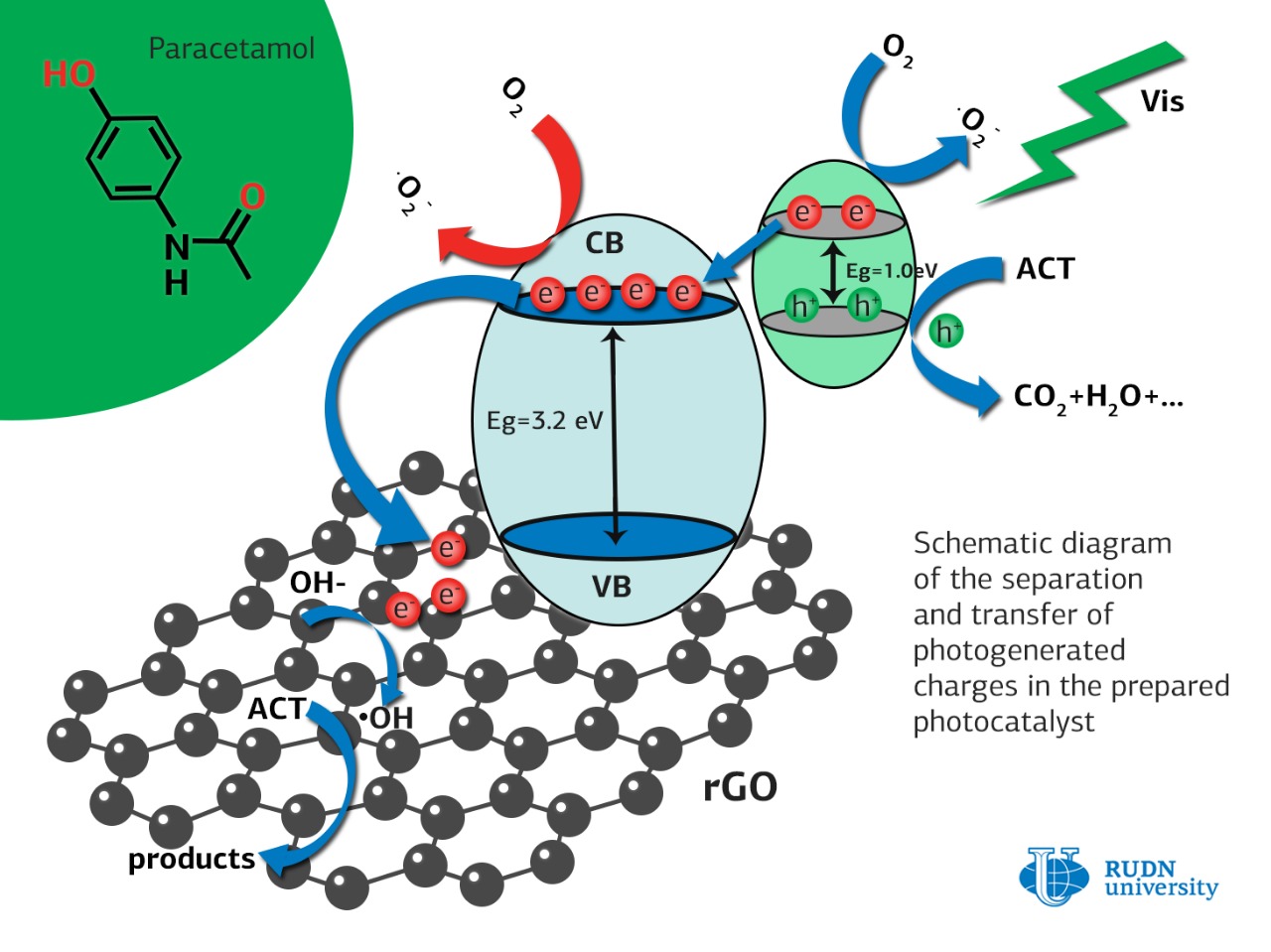
RUDN University chemist Rafael Luque and his colleagues created a series of catalysts for photo-oxidation of acetaminophen in an aqueous medium. The catalyst particles were nanospheres of a composite of zinc oxide and silver sulfide, coated on the outside with a layer of graphene oxide – a layer of carbon one atom thick.
Chemists found that when the catalyst is irradiated with visible light in an aqueous medium, a short-lived and non-toxic superoxide radical is formed from oxygen dissolved in water. It oxidizes acetaminophen to water, carbon dioxide and nitrogen, all harmless to the environment. The sample with 10% molar concentration of silver was the most active.
For comparison, researchers tested the catalytic activity of titanium dioxide and zinc oxide nanospheres coated with graphene oxide in the same experimental conditions. They provided photo-oxidation of acetaminophen at 35 percent for titanium dioxide and 47 percent for zinc oxide in 60 minutes. The new catalysts containing silver sulfide were superior in activity. 100 percent of the acetaminophen disappeared from the solution in 60 minutes.
The study in Separation and Purification Technology
Products derived from microalgae represent a cutting-edge development in the field of bioeconomy. The potential of this biological resource was discussed at the international research seminar “Foundations for a Green Sustainable Energy”, part of the BRICS Network University’s thematic group on “Energy”. The event was organized by the Institute of Ecology at RUDN University.
Ambassadors of Russian education and science met at a conference in RUDN University to discuss how they can increase the visibility of Russian universities and research organizations in the world, and attract more international students in Russia.
The international scientific seminar hosted by RUDN Institute of Ecology “Experience of participation in student organizations as a way to form career skills” united scholarship recipients of the International Student Mobility Awards 2024 and Open Doors, along with members of the scientific student society “GreenLab” and the professional student association “Kostyor (Bonfire)” shared their projects focused on environmental protection.